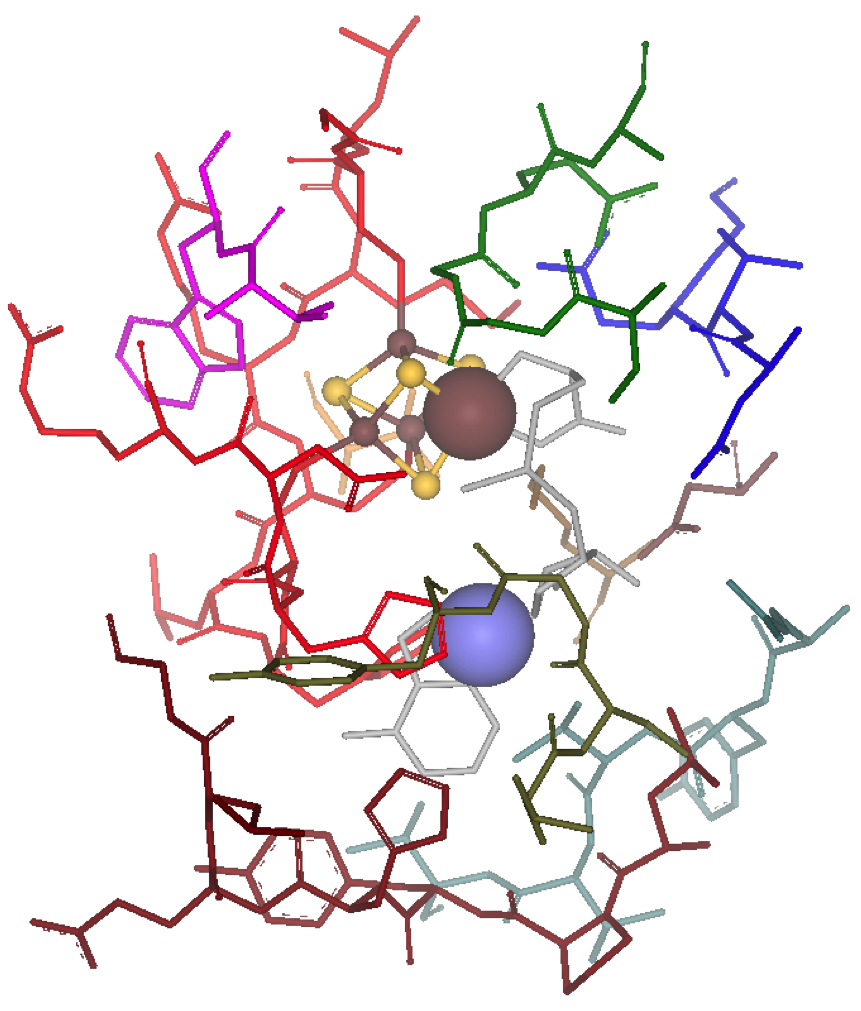
The Fe-S maquette chemistry in our laboratories build on the synergistic application of chemical Synthesis, computational Theory, and analytical Measurements (STM). The students can customize their research training depending on their interests and needs to solve chemical problems, such as defining structure/function relationships of [Fe-S] clusters, exploring their evolutionary role, interrogating their reactivity, and developing new theoretical methods and experimental techniques to study them. Below you can find a few highlights about the current research activities.
Chemical Synthesis
We have established a robust synthetic approach to reproducibly and in high yield prepare [4Fe-4S] maquettes using a wide range of short oligopeptides with varying sequence composition. The as isolated or oxidized form of the maquette is obtained in relatively high yields. The one-electron reduced form, however, persists in lower amounts; our data suggest this is not a simple A -> B one-electron process. All our experiments indicate that the [4Fe-4S]2+ cluster reduction is a one-electron process that is accompanied by cluster decomposition and reassembly process at the [4Fe-4S]+ oxidation level.


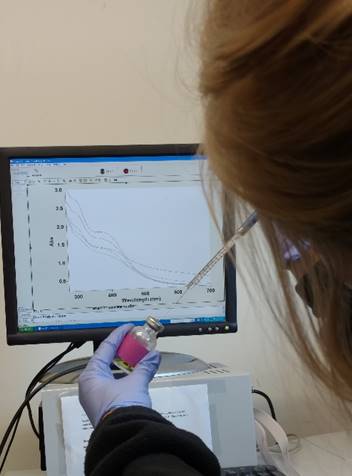

Spectroscopic Characterization
Room temperature UV-Visible spectroscopy is used to monitor the formation of [Fe-S]-maquettes in their as reconstituted or oxidized [4Fe-4S]2+ state. We characterize speciation in the [Fe-S]-maquette complexes via cryogenic electron paramagnetic resonance (EPR) spectroscopy. We are planning future spectroscopic experiments for obtaining structural information at the beamlines of national synchrotron radiation laboratories at Stanford and Berkeley or international laboratories in Japan (Photon Factory).


Computational Chemistry

We take advantage of the full repertoire of computational chemistry tools from empirical force field methods, semi-empirical MO and DFT parameter sets, and ab initio MO and DFT calculations. We utilize spectroscopically validated levels of theory and pay extra attention to realistic representation of solvation in structure, enthalpies, and entropies.
Given the size and composition of the full [4Fe-4S]-peptide maquette, we are developing integrated QM/MM models to describe both the electronic and magnetic structure of the [4Fe-4S] cluster as well as the conformational flexibility of the peptide with or without the cluster present.
Bioinformatics
In collaboration with Prof. Eric Boyd’s GeoBiology laboratory at Montana State University, we investigate the radical SAM metalloenzyme’s cluster binding Cx3Cx2C motif, and compare and contrast it to bacterial ferredoxin Cx2Cx2C motifs to gain insights into the evolution of motif composition and emergence of chemical function. Using the Protein Databank, we map out the most essential intramolecular interactions that define the minimal structural unit to house a redox active [4Fe-4S] cluster coordinated with the SAM cofactor.