Fundamental cellular processes in biology utilize electron transfer events routinely catalyzed by iron-sulfur clusters ([Fe-S]). [Fe-S] clusters are ancient redox active prosthetic groups of metalloproteins and metalloenzymes that exhibit amazing functional diversity. Critical knowledge is lacking in our understanding of the overarching structural and compositional principles that control [Fe-S] cluster redox potential, substrate binding and activation, and [Fe-S] cluster interconversion.
Accordingly, we utilize the versatile synthetic platform afforded by biomimetic maquette chemistry to control and influence structure, composition, and reactivity over a broad range of biological redox potentials. Maquettes are small peptides containing cysteine thiolates that coordinate [4Fe-4S] clusters.
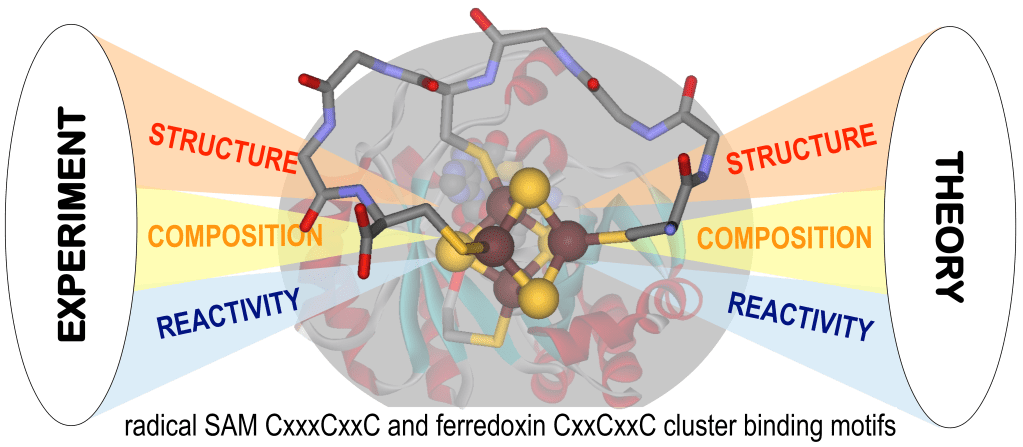
During the first funding cycle, we established robust reconstitution protocols in physiological, buffered conditions, developed analytical spectroscopic techniques for [4Fe-4S]-maquettes, and obtained evidence for site-differentiation, a key requirement for catalysis in [Fe-S]-metalloenzymes.

The driving force for employing maquettes is the hypothesis that [4Fe-4S]- peptide complexes can provide answers to essential questions regarding structure/function relationships of [4Fe-4S]-metalloproteins. We focus on radical S-adenosylmethionine (radSAM) metalloenzymes that couple redox chemistry with bond breaking and formation.
To achieve the aims of the project, we employ techniques from close to twelve orders of magnitude in electromagnetic radiation energy: NMR, EPR, FTIR, UV-Vis, XAS, and Mössbauer spectroscopy, in addition to MS. Interpretation of the experimental results is assisted by computational modeling using empirical force field, semi-empirical Hamiltonians, and ab initio density functional quantum theories. The computational methodologies we develop expected to have broader impacts in metalloenzymology.